 
Keywords: Boyle’s law, P, V, k, pressure of gas, volume occupied by gas, constant, temperature, ideal gas. Calculation of volume of gas: /question/3636135 
Which statement is true for Boyle’s law: /question/1158880Ģ. The graph between the pressure and volume of the helium gas is drawn in the attached image. The temperature of the gas is held constant as the piston is pulled outward. Higher the pressure of the gas, lower will be volume occupied by the gas and vice-versa. 50 A rigid cylinder with a movable piston contains a sample of helium gas. What is the new pressure of the has if the temperature is held at 25 degrees C a. Which graph represents the relationship between the volume of the gas and the pressure of the gas a. Then the has is compressed to a volume of 80mL. A rigid cylinder with a movable piston contains a sample of helium gas. 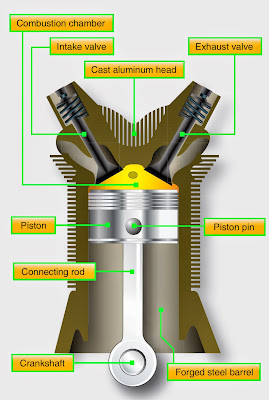
It states that the volume of the gas is inversely proportional to its pressure if the temperature and the number of moles of gas remain constant. At 25 degrees C, gas in a rigid cylinder with a movable piston has a volume of 145mL and a pressure of 125 kPa. Graph of the relationship between pressure and volume of gas. The temperature of the gas is kept constant. But gases tend to behave almost ideally at a higher temperature and lower pressure. It is just a theoretical concept and practically no such gas exists. For the graph, refer to the attached image.Īn ideal gas is a hypothetical gas that is composed of a large number of randomly moving particles that are supposed to have perfectly elastic collisions among themselves. (The boiling point of O2() is – 183☌.The graph that shows the relationship between the volume and pressure of the gas is. Explain why the measured volume of the O2(g) is smaller than the calculated volume. Assume all equipment is functioning properly. (d) The student further cools the cylinder to - 180☌ and observes that the measured volume of the O2(8) is substantially smaller than the volume that is calculated using the ideal gas law. Using principles of kinetic molecular theory, explain why the volume of the 02(8) decreases when the temperature decreases to -55☌. (c) The student tries to change the density of the O2(g) by cooling the cylinder to -55☌, which causes the volume of the gas to decrease. Will this action change the density of the gas remaining in the cylinder? Justify your answer. A rigid cylinder with a movable piston contains a sample of helium gas. The temperature of the gas remains constant at 25☌. 
(b) Attempting to change the density of the O2 (s), the student opens the valve on the side of the cylinder, pushes down on the piston to release some of the gas, and closes the valve again. Calculate the density of the O2(g), in g/L, under these conditions. (a) The cylinder has a volume of 7.95 L at 25☌ and 1.00 atm. The cylinder contains 0.325 mol of 02(8). A student investigates gas behavior using a rigid cylinder with a movable piston of negligible mass, as shown in the diagram above. Transcribed image text: Movable Piston Valve 02(8) 7. a rigid cylinder with a movable piston contains a sample of gas. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
